The how and why of investing in the pharmaceutical sector

Archived article: Please note that tax, investment, pension and ISA rules can change and the information and any views contained in this article may now be inaccurate.
Could investing in pharmaceutical companies get your portfolio in better shape? The pharma sector is, at the very least, difficult to ignore given it represents around a fifth of the MSCI World index. It has also outperformed this global benchmark index over the last 30 years.
In the last three and five years though, pharma stocks have significantly lagged the global market, which has been led by the ‘Magnificent Seven’ and AI-related names.
The sector has also faced headwinds from higher interest rates, rising costs from supply chain issues plus uncertainty on drug prices and, more recently, tariffs.
The pharmaceutical industry is seen as relatively defensive due to the fact people require medical care regardless of economic conditions and government spending on healthcare has increased over time.
There is healthy representation for the pharma sector in the London market. AstraZeneca is the largest company in the FTSE 100 index, with its peer GSK also in the top 10 list of constituents. So, anyone with broad-based exposure to UK stocks will have material exposure to this sector.
Understandably some investors might be wary of putting their money to work in the pharmaceutical space because they might lack the specialist medical knowledge required to fully understand the science and evaluate the risks and opportunities in front of individual businesses.
Fortunately for non-specialists, there are ways to get diversified access through ETFs and actively managed funds, which we discuss later in the article.
What is the outlook for the sector?
The global pharmaceutical sector is a $1.75 trillion industry characterised by rapid technological innovation.
The market is forecast to grow at a compound annual growth rate of between 6% to 8% to reach more than $3 trillion by 2035 with emerging markets showing the fastest growth.
It benefits from structural drivers such as increasing demand for new drugs and therapies related to an aging global population. The World Health Organisation estimates the proportion of people aged over 60 years will nearly double between 2015 and 2050 to 22% from 12%.
Meanwhile the number of people aged 80 or over is expected to triple by 2050 to reach 426 million. While the shift began in high income countries like Japan, by 2050 80% of older people will live in low and middle-income countries.
These trends contribute to the increasing prevalence of chronic disease like cancer, diabetes, obesity, cardiovascular conditions and neurological disorders.
Cancer treatments remain the largest therapy segment with annual revenues expected to top $275 billion in 2025, growing by low double-digit percentages. Autoimmune and inflammatory diseases are the second largest area worth around $175 billion.
Metabolic diseases, especially diabetes and obesity are another high growth segment being fuelled by GLP-1 (Glucagon-like peptide-1) weight-loss drugs which mimic the body’s natural hormone to regulate blood sugar and appetite.
Danish-based Novo Nordisk and US listed Eli Lilly have carved out a significant lead in the obesity market with their respective weekly injectable treatments Wegovy and Zepbound, which remain the only regulated products on the market.
Analysts project combined sales of Wegovy and Zepbound could top $60 billion in 2025 and Morgan Stanley projects the obesity market could be worth $150 billion by 2035.
Morgan Stanley’s Terrance Flynn writes: “We believe we are now at an inflection point for the broadening of obesity drugs’ use, which will extend beyond the U.S. to larger numbers of patients globally.”
We discuss the obesity market in more detail later in this article.
How does the pharmaceutical industry work?
Drug companies are unusual in the business world because their products only have a few years to generate revenues and make a profit before they face stiff competition from generics.
Generics are copycat versions of branded drugs and are usually sold at a cheaper price, reducing demand for the branded version.
Firms look to protect their intellectual property and new inventions by filing patents, which grant exclusivity, typically for a period of 20 years. Patents tend to be filed in the earlier stages of research to prevent a competitor from discovering the idea.
This means the clock starts ticking on the 20-year exclusivity period a long time before a drug candidate has been proven to work and tested for safety through clinical trials.
It can take between eight and 12 years to get a drug approved by a regulator and ready for market, which means the commercial opportunity window is a lot less than 20 years.
Developing drugs is not only time consuming, but also very expensive and highly uncertain. According to the Tufts Centre for the Study of Drug Development, it can cost between $2.2 billion to $2.8 billion to bring a drug to market.
Data suggests only one in 10,000 compounds in pre-clinical research reach the market and around 10% of drugs entering clinical trials get regulatory approval. Half of the drugs which reach late-stage phase three trials go on to be get approved.
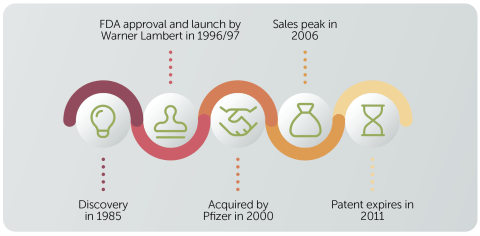
Let’s look at a real example of a drug’s journey to market.
Pfizer’s blockbuster cholesterol drug Lipitor took around a decade to be researched, tested and approved. The drug achieved peak annual sales of $12.9 billion in 2006, and in 2011 the patent finally expired.
During its life as a patented drug Lipitor generated more than $130 billion in worldwide sales, making it the best-selling drug of all time. Sales are estimated to have totalled around $172 billion by the end of 2022.
Lipitor was discovered by Warner-Lambert which Pfizer acquired in 2000 for $90 billion, largely for Lipitor’s potential, standing out as a rare example of a company buying its way to success.
What is driving the increase in mergers and acquisitions?
Due to the unique way the industry operates, it is important for companies to develop a strong pipeline of new drugs to replace the lost revenue from those moving off patent.
Analysts call this risk a patent cliff. Big pharma companies face a collective $236 billion patent cliff between 2025 and 2030 according to consultancy Deloitte, as patents on blockbuster drugs are set to expire.
Analysts project the top 10 firms could see a two-thirds decline in revenues from their key drugs by 2030 without replacements from the pipeline, which explains the elevated number of acquisitions and licensing deals in 2025 which are running at roughly $70 billion.
Examples include Johnson & Johnson’s $14.6 billion purchase of Intra-Cellular and Merck’s $10 billion acquisition of Verona.
What are generics and why do they benefit from patent expiries?
The global generics market is estimated to be worth around $430 billion in 2025 and projected by analysts to grow to $684 billion by 2030.
A key component of growth is expected to come from the increasing number of big-name drugs moving off patent over the next five years.
An adjacent market to generics is biosimilars which refers to drugs based on living cells rather than chemical compounds. Unlike generics, biosimilars are not exact copies of branded drugs due to the complexity and variability of biology.
The US is the largest consumer of generic drugs where over 90% of prescriptions are for generics. India is the often referred to as the ‘pharmacy of the world’ due to its large production and export of generic drugs.
The largest quoted generics companies include Swiss firm Sandoz, Teva, Viatris, Dr. Reddy’s, Fresenius and India’s largest pharma company Sun Pharma. In the UK, generics specialist Hikma Pharmaceuticals is a constituent of the FTSE 100.
What impact are tariffs having?
With the caveat that the US tariff landscape can change without much notice, the current state of play is a tariff framework which sets a ceiling of 15% for imported pharmaceuticals from the EU and Japan.
The US administration announced an executive order to create ‘Most Favoured Nation’ pricing for US patients, with the goal of reducing prices to the lowest priced drugs in other developed countries.
The world’s biggest drug makers operating in the US must offer MFN prices to every single Medicaid patient, the low-income state insurance programme.
Eli Lilly and Pfizer have inked deals whereby they get a three year pause on tariffs in return lowering certain drug prices to the government. In addition, Lilly said it would invest at least $27 billion while Pfizer said it would spend $70 billion to build factories in the US over the next few years.
AstraZeneca has secured a three-year tariff exemption in return for committing $50 billion in US manufacturing and research and development facilities.
Meanwhile, GSK said it will make a $30 billion investment over the next five years in a similar move.
What about China?
The tariff situation is more nuanced in China because while it does not export many finished pharmaceutical products, it is often a monopoly supplier of APIs (Active Pharmaceutical Ingredients) and intermediates needed to manufacture many generic drugs globally.
Some estimates suggest up to 70% of all APIs used in US generics are sourced from China which, if levies are passed on, could lead to higher prices for patients.
To mitigate the risk, companies are busily diversifying their sourcing to other countries. This is forcing a large scale, expensive global realignment of global production.
How big could the obesity market become?
The US is the biggest obesity market in the world with around eight million patients. It is projected to rise to 30 million patients by 2035, representing a penetration rate of between 20% to 25% of the obese population.
The growth prospects for the rest of the world are even greater with the rate of obesity drug penetration estimated to jump from 1% to 10% over the next decade.
Early data on drug launches outside the US suggest high demand in Brazil and India despite the fact patients must cover the cost themselves.
Morgan Stanley equity analyst Thibault Boutherin says: “We anticipate manufacturers’ focus on Asia’s obesity market will grow, particularly in China and Japan, where penetration is low, and innovation continues to emerge.”
Wegovy and Zepbound enjoy currently high market shares but that could change as the market shifts to next generation drugs which offer fewer side effects and convenience.
So far, the excitement around oral pills has not been backed up by the science with both Novo Nordisk and Eli Lilly revealing disappointing clinical trial results in 2025.
Novo is likely to be the first company to launch an oral weight-loss pill given it has already submitted a New Drug Application in the US with approval expected by the end of 2025 or early 2026.
In clinical trials patients taking the daily pill lost around 14% of their bodyweight over 64 weeks.
Eli Lilly’s oral pill Orforglipron has a different chemistry to the injectable version and in clinical trials patients lost an average 11% of their bodyweight on the highest dose.
The company expects to apply to the US regulator before the end of 2025 with potential approval in 2026.
Pfizer discontinued the development of its oral GLP-1 drug candidate Danuglipron in April 2025 after a patient reported a potential drug-induced liver incident.
The company subsequently agreed to purchase weight-loss start up Metsera for up to $10 billion on 12 November after a short-lived takeover battle with Novo Nordisk.
Both companies were attracted to Metsera’s weight-loss drug pipeline which includes a monthly injectable GLP-1 that induced 14% weight loss in mid-stage clinical trials.
The company also has an oral GLP-1 in early development. Metsera shares have risen 167% since their IPO (initial public offering) in January 2025.
Looking to shore up its obesity pipeline Novo struck an exclusive $2.2 billion collaboration deal with San Francisco-based biotech firm Septerna in May 2025.
Other large pharmaceutical groups developing GLP-1s include AstraZeneca and Amgen while smaller biotechnology companies Viking Therapeutics, Zealand Pharma and Structure Therapeutic are also developing weight-loss therapies.
In the private markets space, there are an estimated 157 clinical assets being investigated to combat obesity, 43% of which are aimed at the oral pill market with seven in late-stage clinical trials, according to data from industry research specialist IQVIA.
What is biotechnology?
Biotechnology companies focus on biological processes such as genetic engineering and cell research to create ‘biologic’ drugs to improve human life. Whereas pharmaceutical firms tend to concentrate on developing and manufacturing drugs through chemical synthesis, often with more established process and more consistent streams of revenue. The biotech field also includes areas like agriculture and environmental science.
Biotechnology companies are often smaller pre-revenue firms, focused on research and development, which makes them riskier investments.
Share prices can move around a lot, both up and down, especially around the release of clinical trial results.
Biotech companies often rely on external funding from venture capital firms or partnerships with larger pharmaceutical firms to sustain their research and development.
The most used benchmark tracking quoted biotechnology shares is the NBI or Nasdaq Biotechnology Index.
Over the last five years, the NBI has lagged the broad S&P 500 index by a wide margin, gaining just 25% compared with the near doubling for the S&P 500 index.
However, since May 2025 the NBI has seen a significant rally, gaining 37% compared with 17% for the S&P 500 index.
This has been driven by an increase in M&A (mergers and acquisition) activity and the IPO market showing signs of life after many years in the wilderness.
Indicative of rising investor enthusiasm was molecular diagnostics firm BillionToOne whose shares briefly touched $120 on their first day of dealings on Nasdaq after listing at just $60 per share.
Ways to get passive exposure to the global pharmaceutical sector
The largest and cheapest ETF is the Xtrackers MSCI World Health Care UCITS ETF which has £2.5 billion of assets under management.
The fund seeks to replicate the performance of the MSCI World Health Care index by buying all the index constituents. Dividends are accumulated and reinvested in the ETF.
Around two thirds of the fund is invested in US stocks and top holdings include Eli Lilly, Jonhson & Johnson, AbbVie and AstraZeneca. The ETF has an annual charge of 0.25%.
The largest and cheapest ETF tracking the Nasdaq Biotechnology index is the iShares US Nasdaq Biotechnology UCITS ETF which has just over half a billion pounds in assets under management.
The ETF holds over 250 names and seeks to replicate the performance of the NBI by buying a selection of the most relevant index constituents. Dividends are accumulated and reinvested.
Around 75% of the fund is invested in US companies and the top 10 holdings represent 45% of total assets.
What about actively managed funds and trusts?
There are specialist actively managed funds and trusts available which aim to give investors diversified exposure to the sector. These vehicles have higher annual charges than ETFs.
The main difference between trusts and funds is that the former, also called closed ended funds, can trade at a premium or discount to the value of the assets in the portfolio.
Investment trusts can borrow money to invest which is not possible for open ended funds. Investment trusts trade like shares while funds are usually priced daily.
The largest trust in the sector is Worldwide Healthcare Trust which has £1.5 billion of assets under management. The trust trades on a 7% discount to net asset value and the annual charge is 0.9% a year.
The fund is managed by Orbimed Advisors, one of the largest dedicated healthcare firms in the world with over $19 billion of assets under management.
The investment team has a global focus and covers the entire spectrum of companies, from early stage to fully integrated biopharmaceutical companies.
The team looks for companies with underappreciated products in the pipeline, and high-quality management teams. Since launch in 1995 the fund has generated an annualised total return in net asset value of 13.4% a year, compared with an index return of 10.9% a year.
The largest actively managed biotechnology fund is Polar Capital Biotech, which has £1.9 billion of assets under management and an annual charge of 1.1%. The fund typically holds between 40 and 60 positions.
Managed by a team of eight sector specialists with a combined 150 years of industry experience, the fund looks to invest in cutting edge medical innovation where the team believes the stock market is underappreciating a company’s potential.



